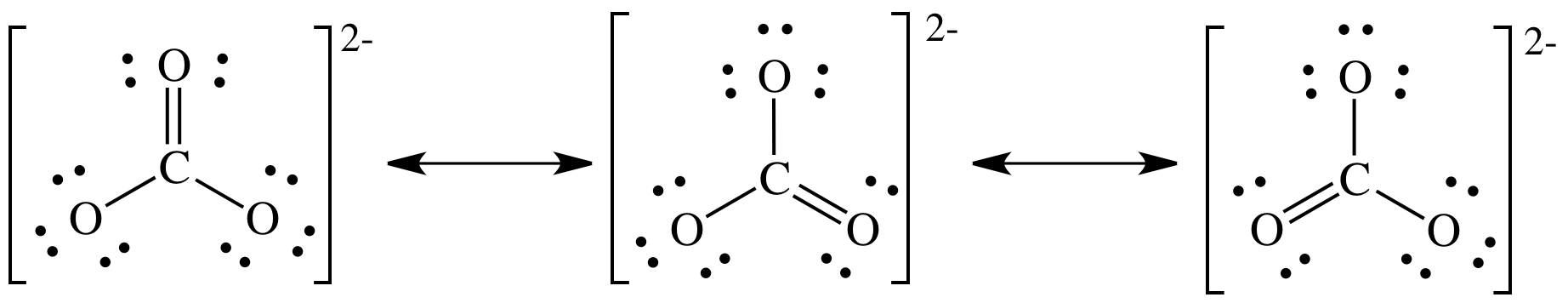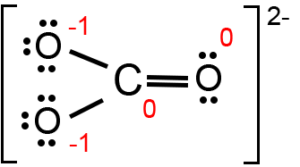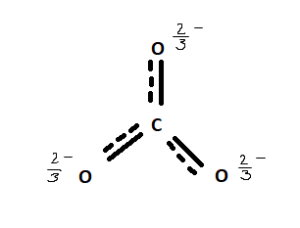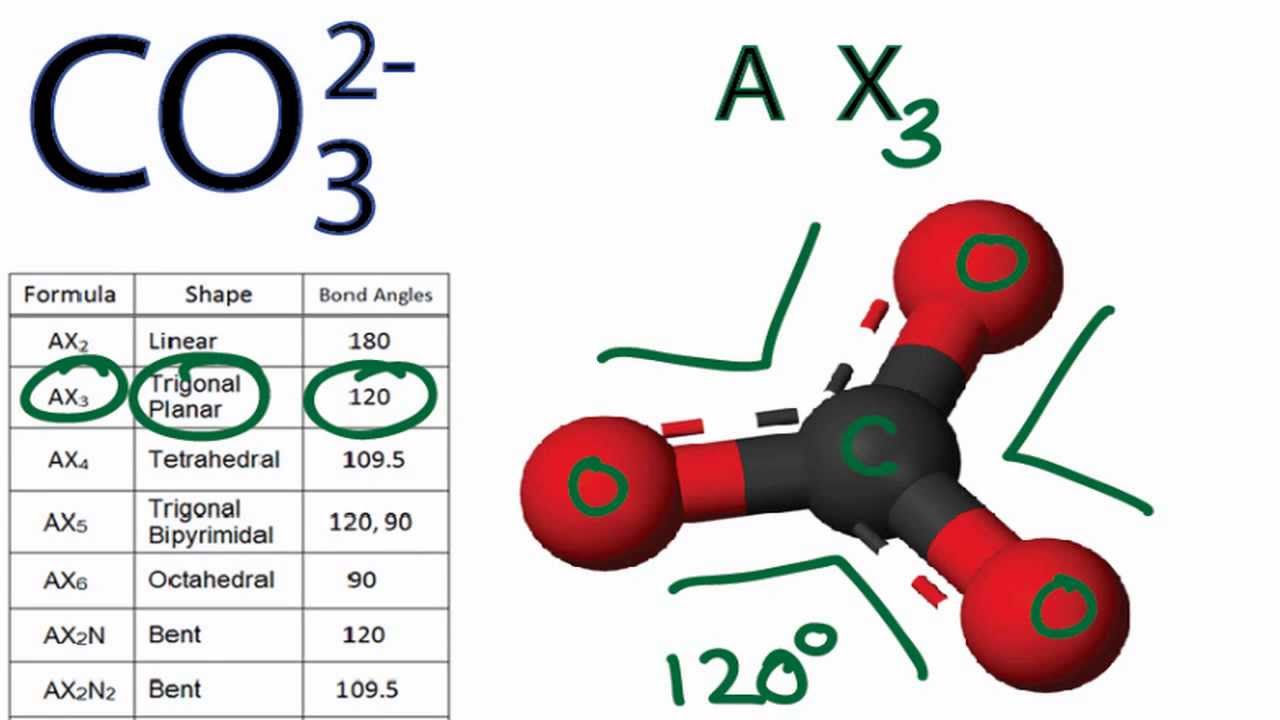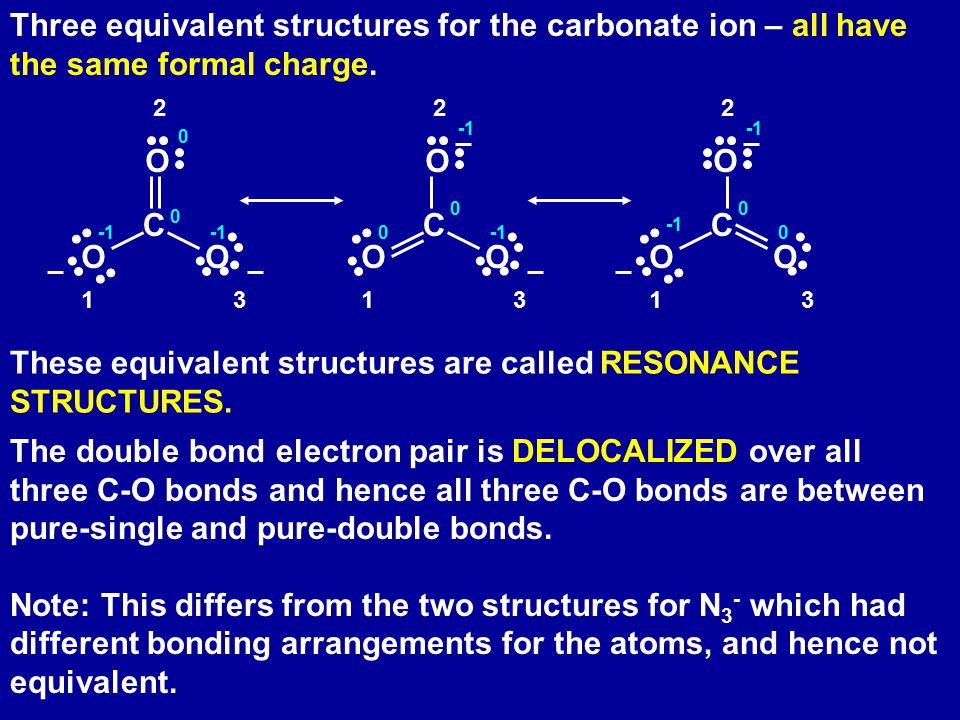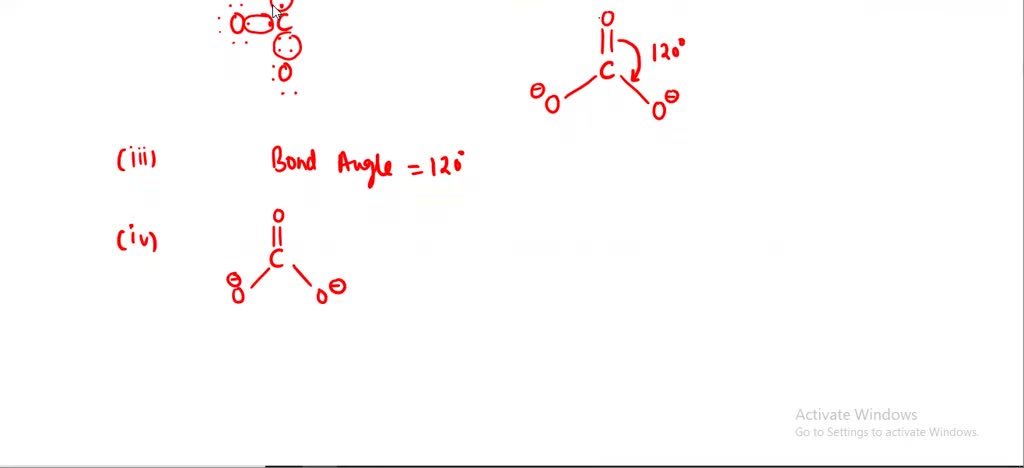
SOLVED: Draw the lewis structure of CO3^2- 1. give the predicted structure for CO3^2- based on VSEPR 2. give the hybridization for the central atom in CO3^2- 3. give the bond angle(s)

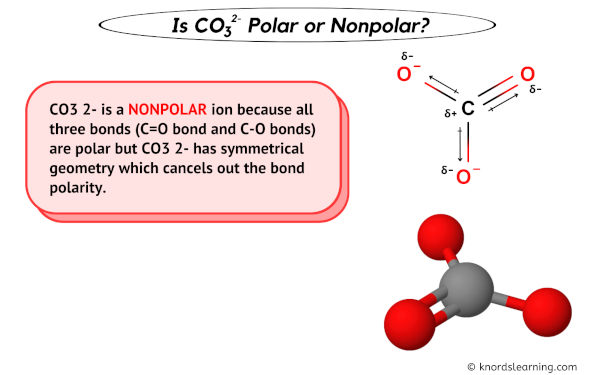
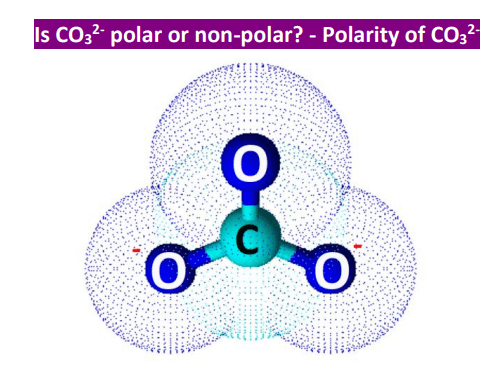
Geometry Of Molecules - Is CO3 2- Polar or Nonpolar (Carbonate ion) | Facebook | By Geometry Of Molecules | Is CO3 2- Polar or Nonpolar (Carbonate ion) Carbonate ion is a
Draw a Lewis structure for CO32- and answer the following questions. a. What is the number of lone pairs? b. What is the number of single bonds? c. What is the number

Determine if each compound or ion below has a dipole moment. (a) Carbonate ion (CO_3 ^{2-}) | Homework.Study.com
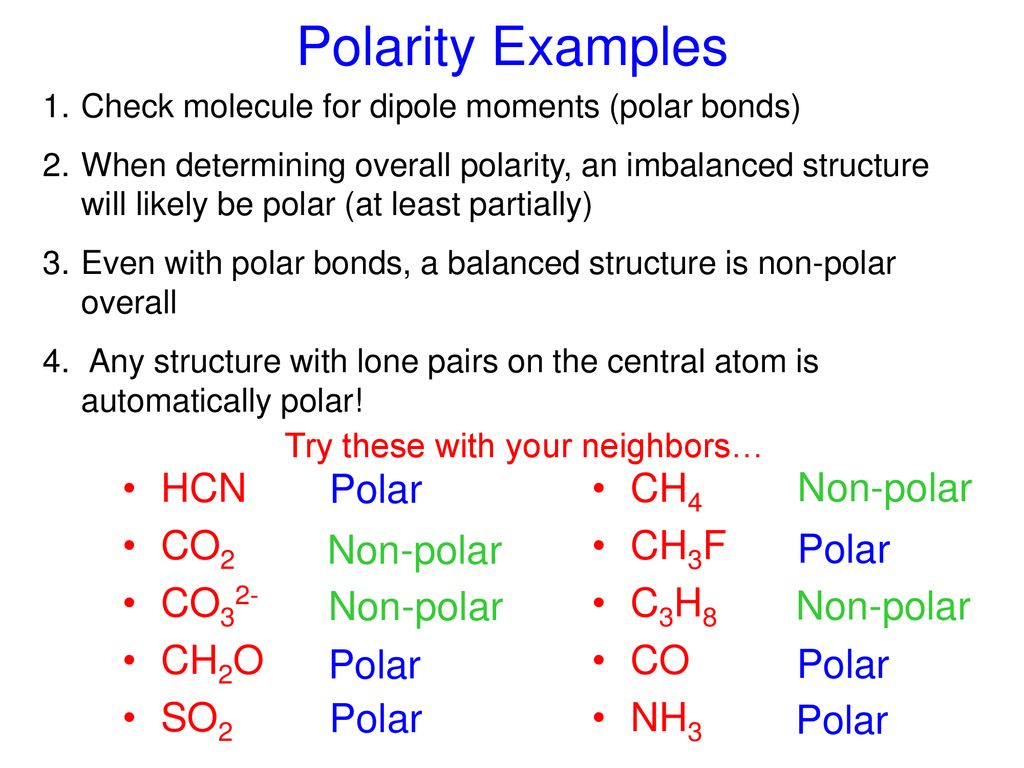
amongst NO3^ 1, AsO3^ 3,CO3^ 2, ClO3^ 1, SO3^ 2 and BO3^ 3, the non polar species are: (1) NO3^ 1,CO3^ 2, BO3^ 3 (2) AsO3^ 3, ClO3^ 1, SO3^ 2 (3) CO3^ 2, SO3^ 2, AsO3 3 (4) ClO3^ 1, SO3^ 2, NO3^ 1

Can a molecule (like CO3 2-) be nonpolar even if it has two single bonds and one double bonds? - Quora