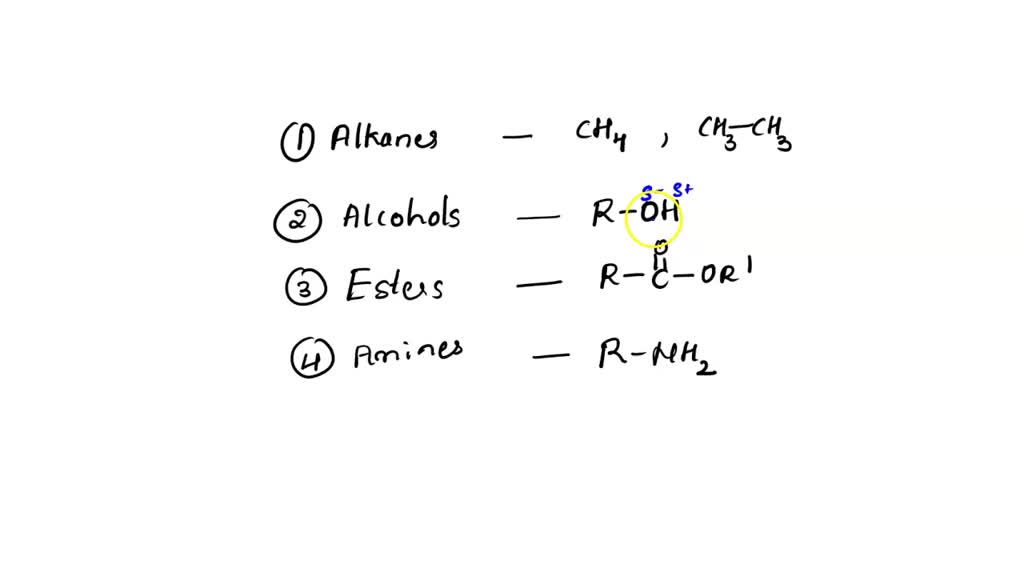
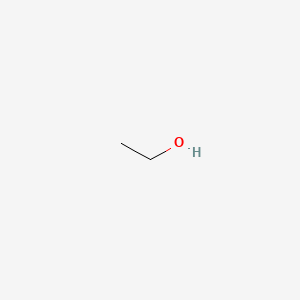
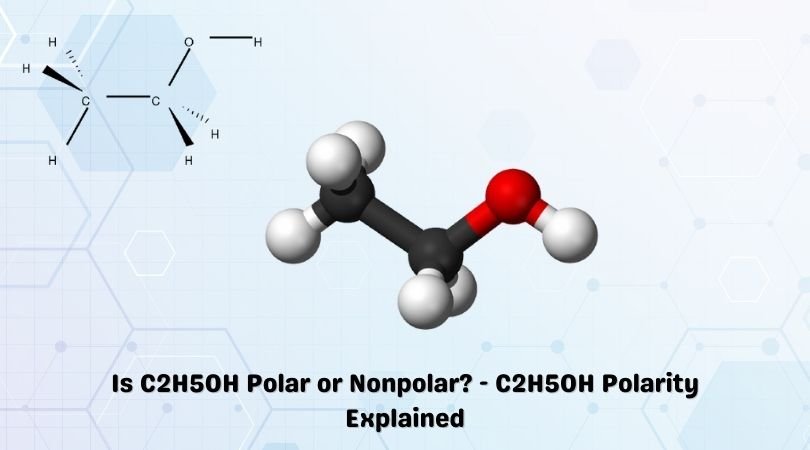
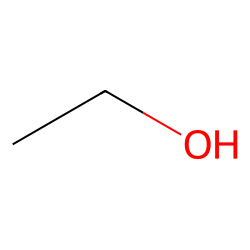
C2H5OH Lewis structure, molecular geometry, hybridization, bond angle | Molecular geometry, Molecular, Electron configuration

Growth Pattern, Stability, and Properties of Complexes of C2H5OH and nCO2 (n = 1–5) Molecules: A Theoretical Study | ACS Omega

Determine whether the molecule CH_2CHCH_3 is polar: A) The molecule is polar only because there are slightly polar bonds. B) The molecule is polar because there are slightly polar bonds and the
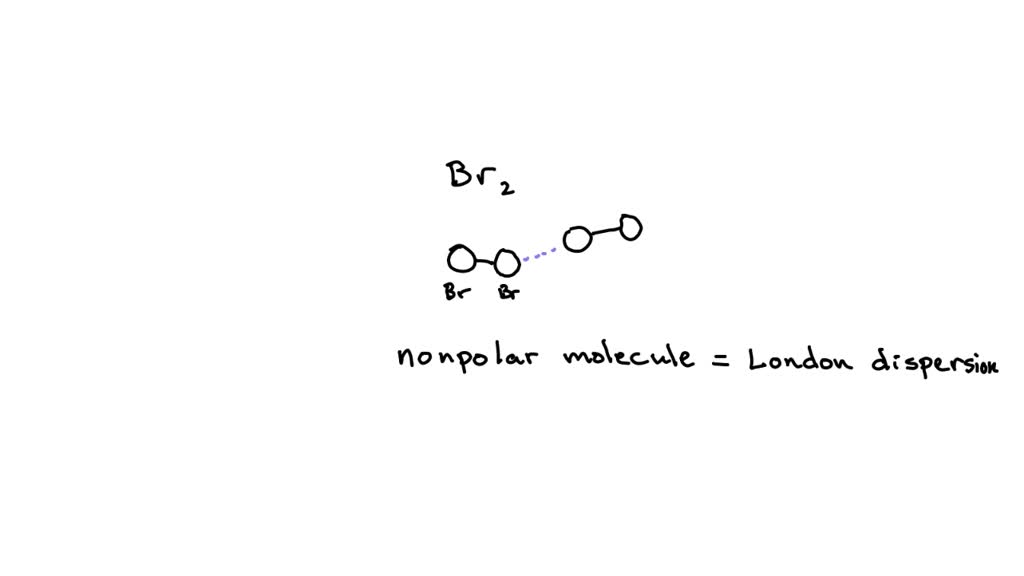
SOLVED: What main type of forces must be overcome between Br2 molecules when liquid Br2 dissolves in C2H5OH ethanol, ? What main type of forces must be disrupted between C2H5OH molecules when

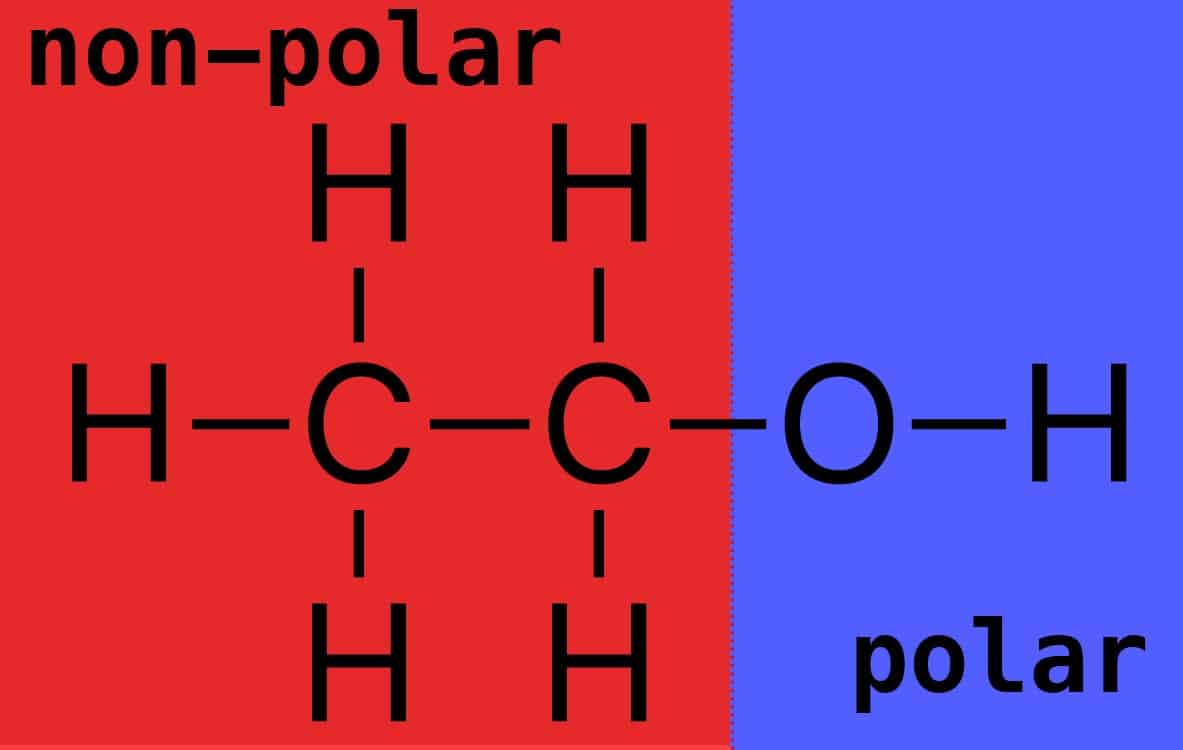
What intermolecular forces are present in C2H5OH? a. Dispersion forces only b. Dispersion forces and dipole-dipole forces c. Dispersion forces, dipole-dipole forces, and hydrogen bonding | Homework.Study.com
![PDF] Measurement and Modeling of Carbon Dioxide Solubility in Polar and Nonpolar Solvent | Semantic Scholar PDF] Measurement and Modeling of Carbon Dioxide Solubility in Polar and Nonpolar Solvent | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4e1fdbc8b08394d57fd8a65ca57454ec1bcfb94d/2-Table1-1.png)
PDF] Measurement and Modeling of Carbon Dioxide Solubility in Polar and Nonpolar Solvent | Semantic Scholar